教授, FRSC
深圳研究生院化学生物学与生物技术学院
叶涛于1993年在女王大学获得有机化学博士学位。并在女王大学继续进行博士后研究工作。他于1994年在诺丁汉大学出任ROPA fellow,然后任教于香港大学 (1998-2001),香港理工大学 (2001-2015)。 叶涛教授自2015年9月全职回到北大深圳研究生院工作, 负责化学生物学及生物技术学院的学术委员会工作,目前兼任学院的常务副院长。
RESEARCH INTEREST
Our research interests span the disciplines of natural product synthesis, chemical biology and drug discovery, which include the discovery and development of new agents of medicinal value through major advances in chemical synthesis. Currently, we are working in two main areas:
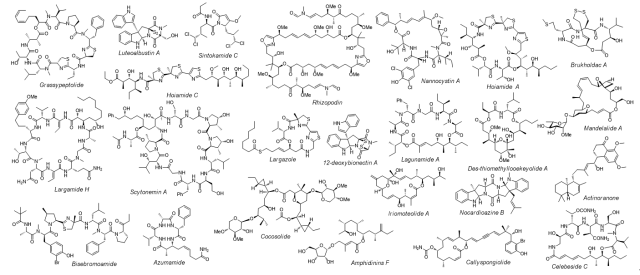
1. Total Synthesis and Biological Evaluation of Natural Products and Their Analogues. Natural products have provided considerable value to the pharmaceutical industry over the past half century. In particular, the therapeutic area of oncology has benefited from numerous drug classes derived from natural product sources. Chemical synthesis of natural products and their analogues has been a key tool in drug discovery and development. The synthesis allows verification of primary structure proposed on the basis of studies of natural product, and presents opportunities to modify the structure, with the ultimate aim of improving activity or physicochemical/biological properties of the lead molecule. Synthesis is also crucial in the establishment of structure-activity relationships since the ability to make analogues of the lead compound chemically is a prerequisite of drug discovery. We have been particularly devoting to the exploration of natural-products-based drug discovery. The larger part of our research program is dedicated to the training and research in synthesis and biological evaluation of natural products with known biological activities. Structures of some completed molecules are shown below:
2. Design and Syntheses of Novel Small Molecule Bioprobes and Drugs.
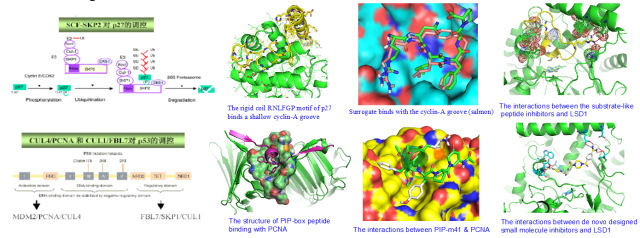
Protein–protein interactions play a key role in most biological processes, and offer attractive opportunities for therapeutic intervention. The targeted manipulation of protein-protein interactions with the use of small molecules is rapidly gaining importance in the development of biological tools for dissecting living processes on a molecular level and for the discovery of conceptually novel drugs. Our research centers on rationally designed molecular probes / drug candidates, and their application to biological problems, especially in cancer biology and neurodegenerative disorders.
SELECTED PUBLICATION
1. Wang, L.; Wu, F.; Jia, X.; Xu, Z.; Guo, Y.; Ye, T. “Studies toward the Synthesis of Iriomoteolide-2a: Construction of the C(6)−C(28) Fragment” Org. Lett. 2018, 20, 2213-16.
2. Chen, K.; Xu, Z.; Ye, T. “Total Synthesis of Amphidinins E, F and epi-Amphidinin F” Org. Chem.Front. 2018, 5, 629-32.
3. Li, L.; Tang, M.; Tang, S.; Gao, S.; Soliman, S.; Hang, L.; Xu, W.; Ye, T.; Watanabe, K.; Tang, Y. “Genome Mining and Assembly-Line Biosynthesis of the UCS1025A Pyrrolizidinone Family of Fungal Alkaloids” J. Am. Chem. Soc. 2018, 140, 2067-71.
4. Lei, H.; Wang, L.; Xu, Z.; Ye, T. “Regio- and Stereospecific Construction of 3a-(1H Indol-3-yl)pyrrolidinoindolines and Application to the Formal Syntheses of Gliocladins B and C” Org. Lett. 2017, 19, 5134-37.
5. Guo, Y.; Zhao, M.; Xu, Z.; Ye, T. “Total Synthesis and Stereochemical Assignment of Actinoranone” Chem. Eur. J. 2017, 23, 3572–3576.
6. Liao, L.; Zhou, J.; Xu, Z.; Ye, T. “Concise Total Synthesis of Nannocystin A” Angew. Chem. Int. Ed. 2016, 55, 13263-13266.
7. Zhou, J.; Gao, B.; Xu, Z.; Ye, T. “Total Synthesis and Stereochemical Assignment of Callyspongiolide” J. Am. Chem. Soc. 2016, 138, 6948-6951.
8. Gunasekera, S. P.; Li, Y.; Ratnayake, R.; Luo, D.; Lo, J.; Reibenspies, J. H.; Xu, Z.; Clare-Salzler, M. J.; Ye, T.; Paul, V. J.; Luesch, H. “Discovery, Total Synthesis and Key Structural Elements for the Immunosuppressive Activity of Cocosolide, a Symmetrical Glycosylated Macrolide Dimer from Marine Cyanobacteria” Chem. Eur. J. 2016, 22, 8158-8166.
9. Liu, J.; Wang, L.; Zhang, J.; Xu, Z.; Ye, T. “The total synthesis and stereochemical assignment of scytonemin A” Chem. Commun., 2016, 52, 1002-1005.
10. Qu, S.; Chen, Y.; Wang, X.; Chen, S.; Xu, Z.; Ye, T. “Total Synthesis of Largamide B” Chem. Commun. 2015, 51, 2510-2513.
11. Lei, H.; Yan, J.; Yu, J.; Liu, Y.; Wang, Z.; Xu, Z.; Ye, T. “Total Synthesis and Stereochemical Reassignment of Mandelalide A” Angew. Chem. Int. Ed. 2014, 53, 6533-6537.
12. Zhang, X.; Lu, F.; Wang, J.; Yin, F.; Xu, Z.; Qi, D.; Wu, X.; Cao, Y.; Liang, W.; Liu, Y.; Sun, H.; Ye, T.; Zhang, H. "Pluripotent Stem Cell Protein Sox2 Confers Sensitivity towards LSD1 Inhibition in Cancer Cell", Cell Reports, 2013, 5, 445-457.
13. Song, L.; Liu, J.; Gui, H.; Hui, C.; Zhou, J.; Guo, Y.; Zhang, P.; Xu, Z.; Ye, T. “Synthesis of the Macrocyclic Core of Rhizopodin” Chem. Asian J. 2013, 8, 2955-2959.
14. Liu, H.; Liu, Y.; Wang, Z.; Xing, X.; Maguire, A. R.; Luesch, H.; Zhang, H.; Xu, Z.; Ye, T. “Total Synthesis and Biological Evaluation of Grassypeptolide A” Chem. Eur. J. 2013, 19, 6774-6784.
15. Long, B.; Tang, S.; Chen, L Qu, S.; Chen, B.; Liu, J.; Maguire, A. R.; Wang, Z.; Liu, Y.; Zhang, H.; Xu, Z.; Ye, T. “Total Synthesis of Padanamides A and B” Chem. Commun. 2013, 49, 2977-2979.
16. Dai, L.; Chen, B.; Lei, H.; Wang, Z.; Liu, Y.; Xu, Z.; Ye, T. “Total Synthesis and Stereochemical Revision of Lagunamide A” Chem. Commun. 2012, 48, 8697-8699.
17. Wang, M.; Feng, X.; Cai, L.; Xu, Z.; Ye, T. “Total Synthesis and Absolute Configuration of Nocardioazine B” Chem. Commun. 2012, 48, 4344 - 4346.
18. Wang, J.; Lu, F.; Ren, Q.; Sun, H.; Xu, Z.; Lan, R.; Liu, Y.; Ward, D.; Quan, J.; Ye, T.; Zhang, H. “Novel Histone Demethylase LSD1 InhibitorsSelectively Target Cancer Cells with Pluripotent Stem Cell Properties” Cancer Research, 2011, 71, 7238-7249.
19. Wang, L.; Xu, Z.; Ye, T. “Total Synthesis of Hoiamide C” Org. Lett. 2011, 13, 2506-2509.
20. Liu, Y.; Wang, Z.; Kwong, S.; Lui, E. L. H.; Friedman, S. L.; Li, F.; Lam, R. W. C.; Zhang, G. C.; Zhang, H.; Ye, T.; “Inhibition of PDGF, TGF-β and Abl Signaling and Reduction of Liver Fibrosis by the Small Molecule Bcr-Abl Tyrosine Kinase Antagonist Nilotinib” J. Hepatology, 2011, 55, 612-625.
Book: “Modern Catalytic Methods for Organic Synthesis with Diazo Compounds: From Cyclopropanes to Ylides” (652 pages), Doyle, M. P.; McKervey, M. A.; Ye, Tao, John Wiley & Sons, Inc., New York. 1998